Audit ready.
Every hour. Automatically.
Pharma compliance software that connects to your systems, scans every hour, and tells you exactly which regulatory clause you're failing before the auditor does.
10x
Faster audit prep
80%
Less manual work
9+
Frameworks covered
25-second product overview · Auto-plays
The Problem
Compliance demands
keep growing.
Manual evidence collection, stale documentation, last-minute audit scrambles. Sound familiar?
Assessment Due
SOC 2 Type II audit due in 14 days
237 controls to validate manually
Gap Detected
21 CFR Part 11 §11.10(e) — no audit trail
Found during last FDA inspection prep
Evidence Expired
AWS CloudTrail evidence is 47 days stale
12 controls affected across 3 frameworks
How It Works
From zero to audit ready
Connect your tools. DueVault auto collects evidence, maps it to 21 CFR Part 11 controls, scores your compliance posture, and generates audit packages on a schedule you set.
Connect
Link GitHub, AWS, Okta 12 tools supported
Collect
Evidence auto collected on schedule (hourly/daily)
Map
Each evidence item maps to specific framework controls
Score
Compliance score updates with trends (improving/declining)
Report
One click audit packages for FDA inspections
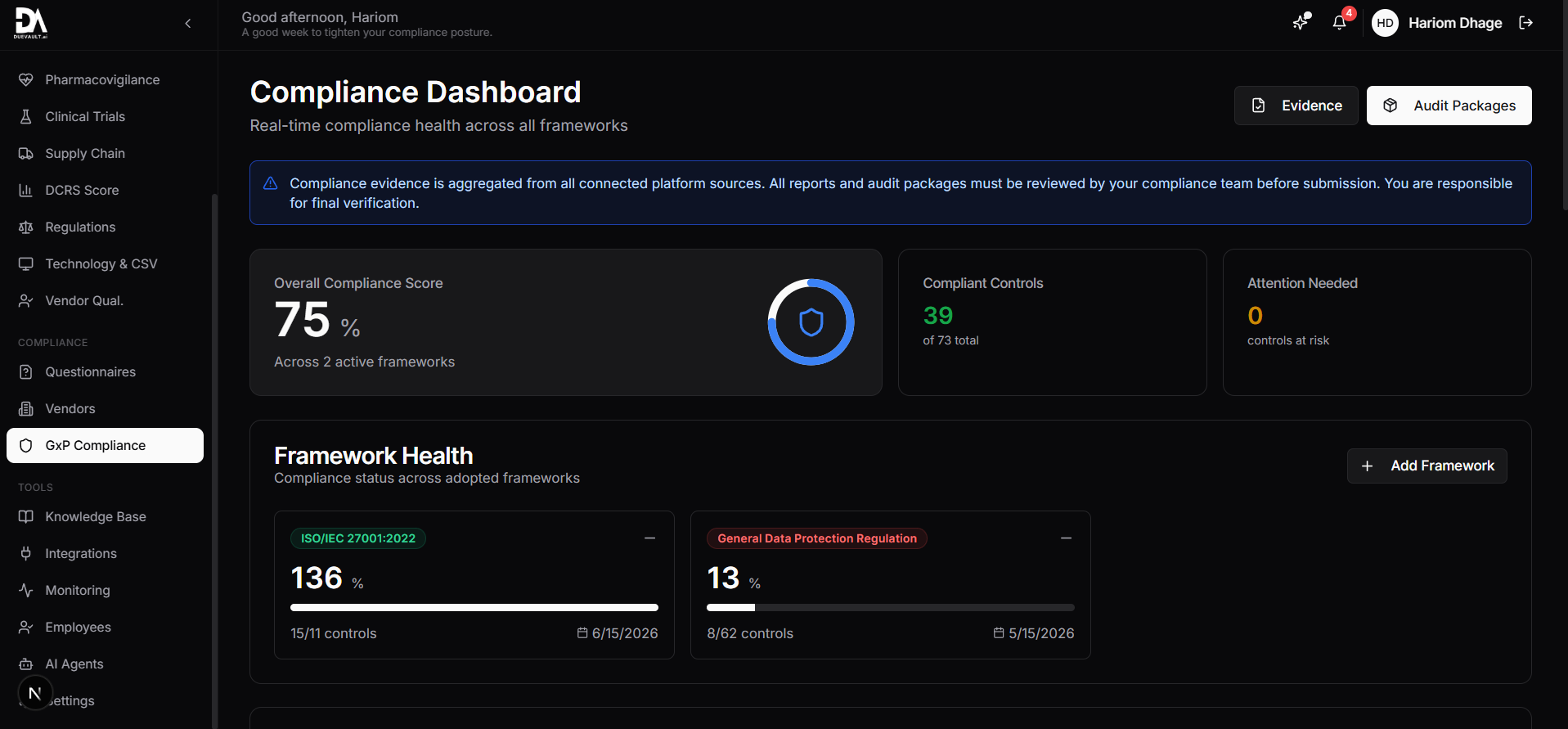
Real time compliance scoring 75% overall, framework health with trend indicators, 39 compliant controls
AI That Does The Work
Ask a question.
Get a complete solution.
4 specialized AI agents that understand pharma compliance. They generate protocols, find gaps, analyze deviations, and prepare audit packages.
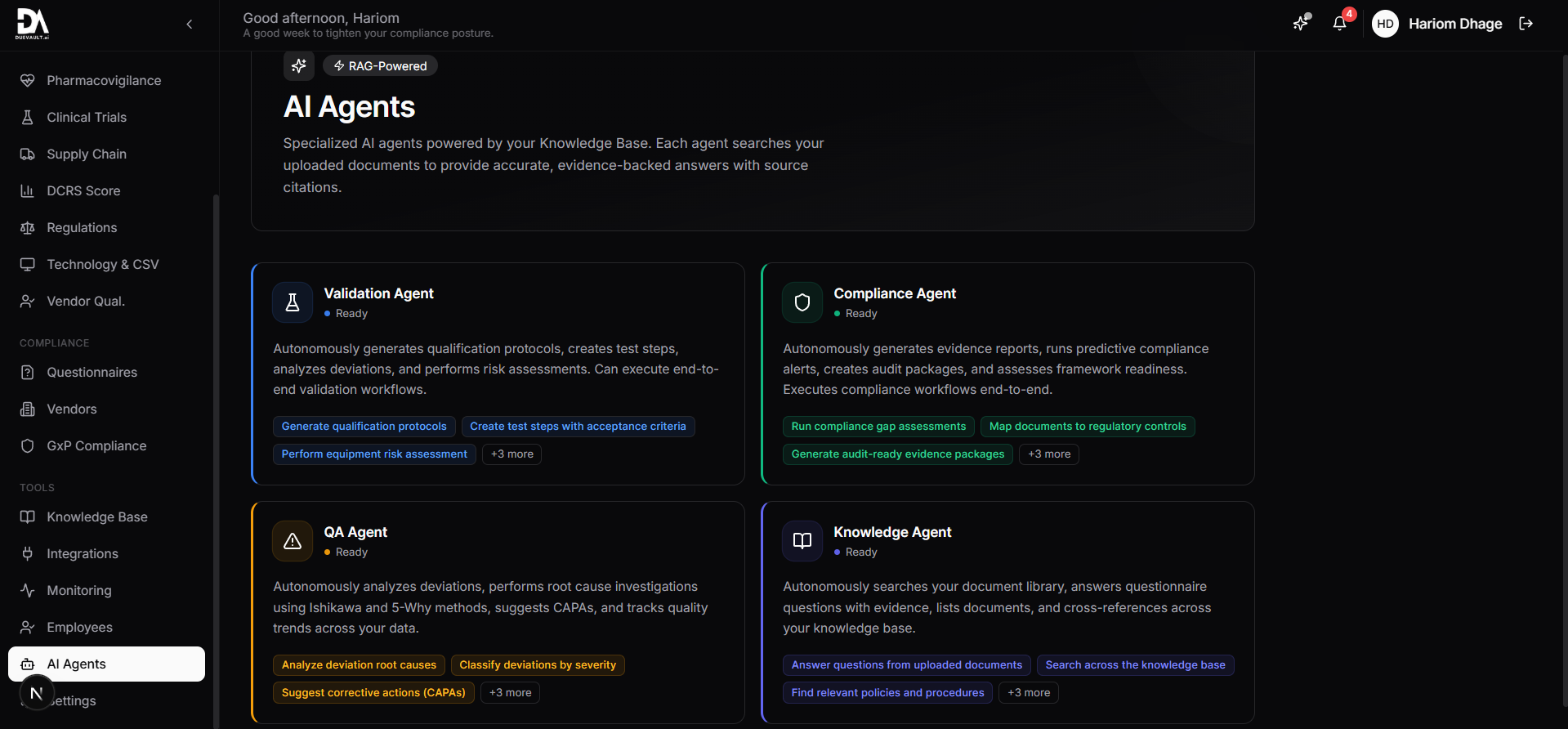
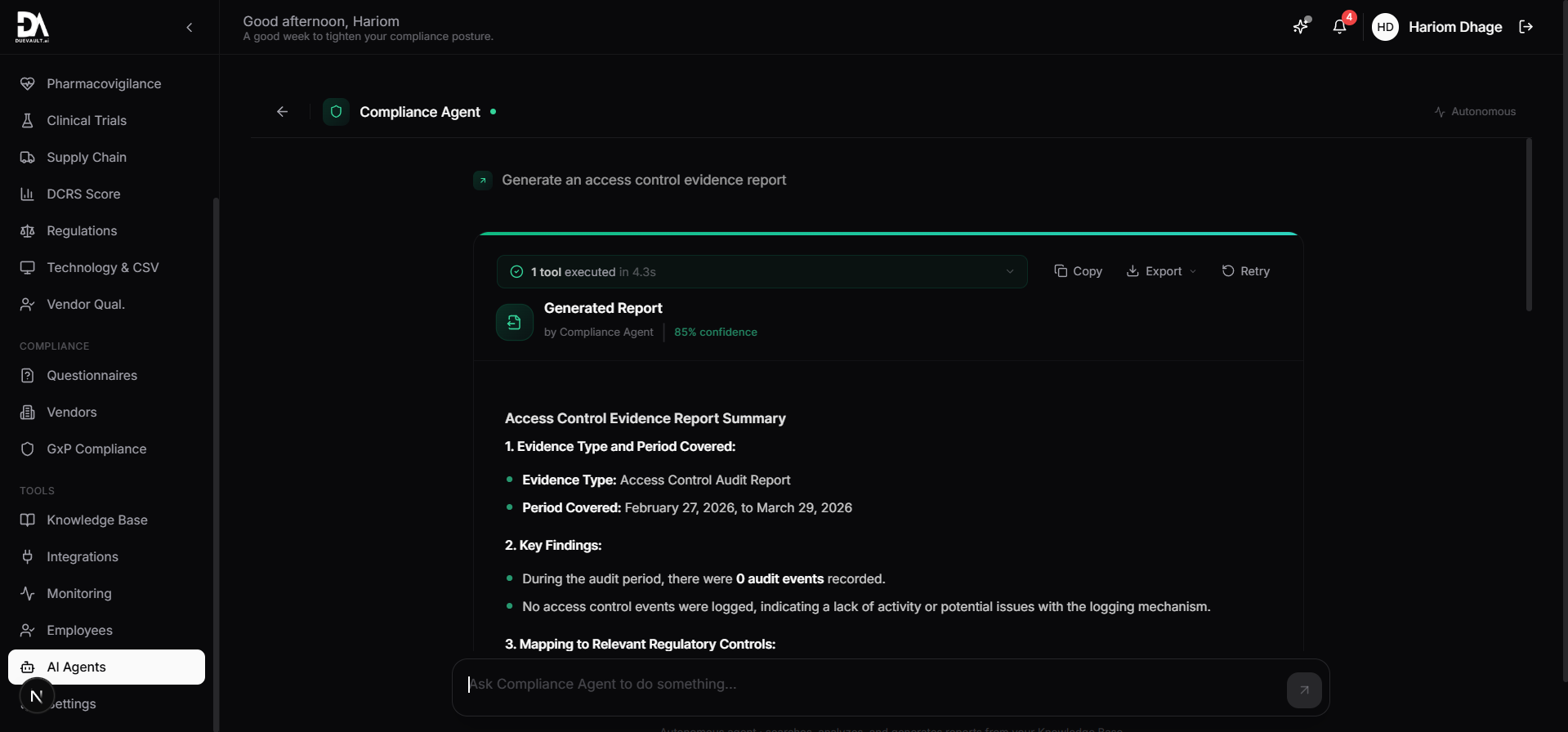
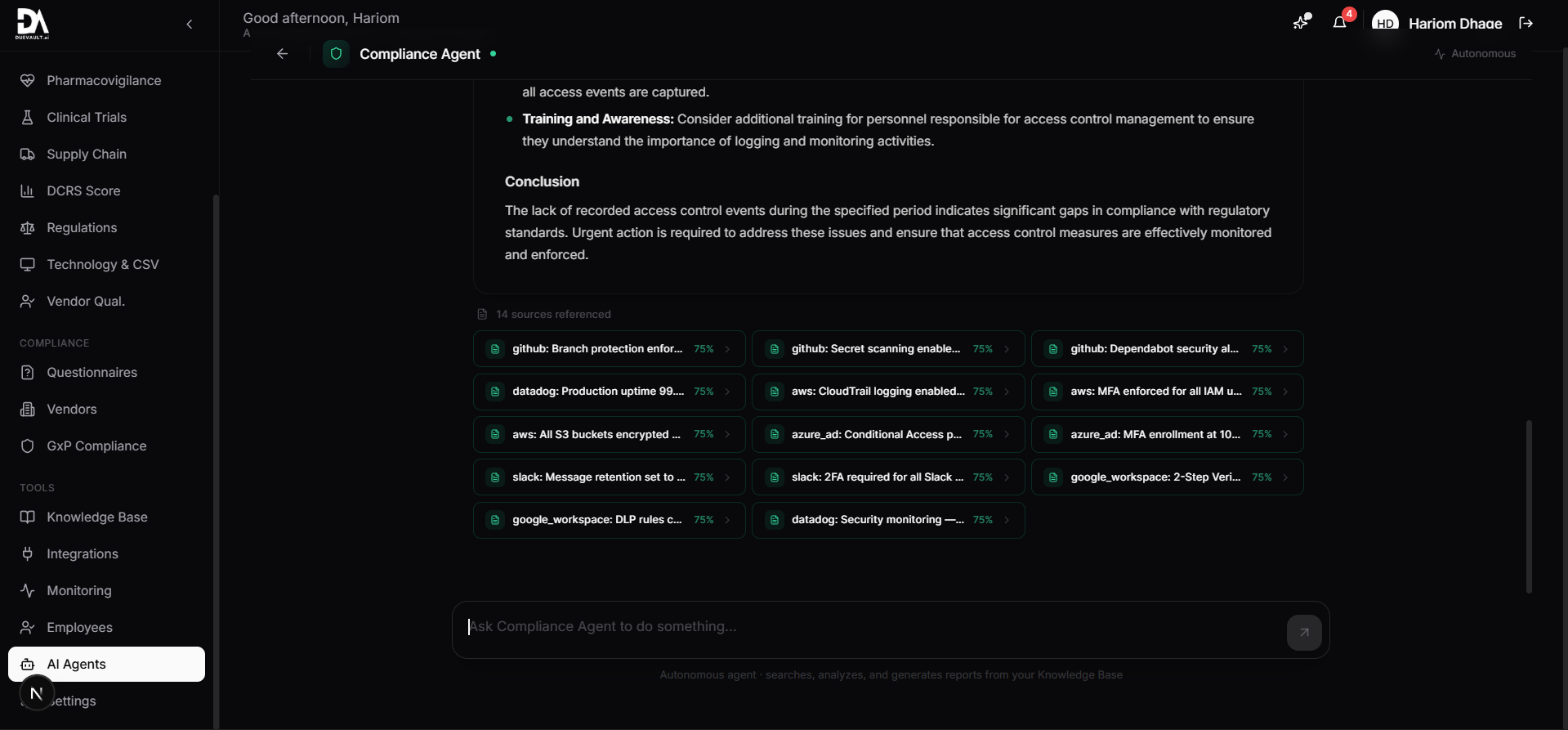

192
Security Policies
Complete data isolation
21 CFR
Part 11 Compliant
From day one
0
Data Retention on AI
Prompts never stored
100%
On Premise Ready
Air gapped deployment
Founded by Dr. Balaji Ommurugan (15+ years at Novartis, Vifor, Organon) and Hariom Dhage
Compliance Frameworks
Questions? We're here to help.